  
The 1/2 in the equation accounts for the fact that bonding electrons are shared between two atoms. The result is the formal charge for the atom.įormal Charge = Valence Electrons – Lone Pair Electrons – 1/2 Bonding Electrons Subtract the number of electrons in step 2 from the number of valence electrons in step 1.To do this, count the number of electrons in the atom’s bonds and lone pairs. Count the number of electrons that each atom has in the molecule.For example, carbon has four valence electrons, nitrogen has five, and oxygen has six. The number of valence electrons for an atom can be found by looking at its position on the periodic table. Find the number of valence electrons for each atom in the molecule.To calculate formal charge, follow these steps: Formal charge is used to determine the most stable arrangement of atoms and to predict the reactivity of a molecule. It is the difference between the number of valence electrons an atom would have if it were isolated and the number of valence electrons it actually has in a molecule. What is Formal Charge?įormal charge is the charge assigned to an atom in a molecule. In this article, we will discuss how to calculate formal charges and provide examples of its application. It can help predict the reactivity of a molecule and the most stable arrangement of atoms. How to Calculate Formal Charge on Atoms in Lewis Structures How To Calculate Formal Chargeįormal charge is a tool used in chemistry to determine the distribution of electrons in a molecule. Simple Tips for Calculating Formal Charge in Chemistryĥ. How to Determine Formal Charge in Ionic CompoundsĤ. 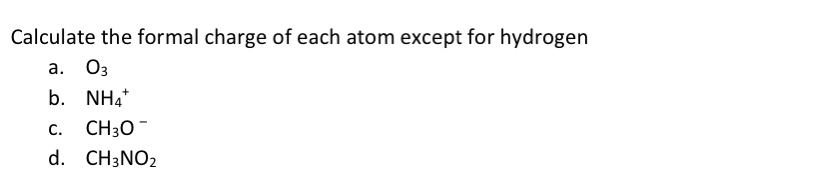
Step-by-Step Guide on How to Calculate Formal Chargeģ. How to Calculate Formal Charge in MoleculesĢ. Calculating Formal Charges: A Guide to Mastering the Basics 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
